Stay up to date
Keep up with the latest products and trends in digital health.
About AVIA Marketplace
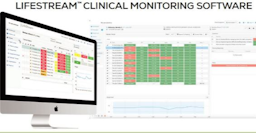
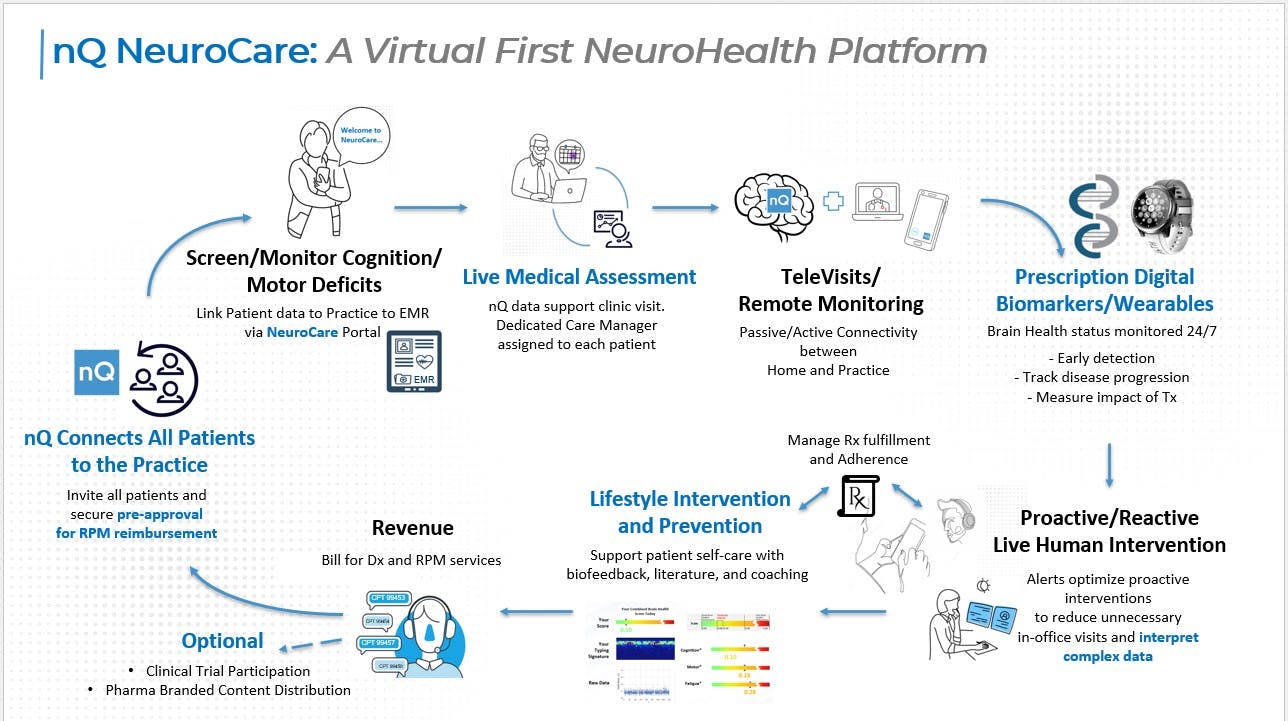
AVIA Marketplace is where innovative health systems and hospitals go to find the right digital health solutions. It’s built to efficiently search vendors and guide informed decisions, with tools like product comparisons, compatibility level, report generators, peer reviews, and market insights.
AVIA Marketplace is brought to you by AVIA, a trusted company and community of experts who guide health systems on their digital transformation journey.
Top Categories